Malaria still affects 200 million people worldwide and kills 600,000 of them - especially children. Scientists have confirmed the existence of malaria parasites (Plasmodium falciparum) in Africa as early as 2021 that have developed resistance to derivatives of artemisinin, the main ingredient in the first-line treatment for malaria.The study was published in the New England Journal of Medicine.(1, 2, 3)
Malaria is a complicated infection process involving several stages: After an Anopheles mosquito transmits the malaria pathogens (sporozoites) while sucking blood, these migrate via the bloodstream to the liver, infecting the liver cells. In the liver, they mature into schizonts and release merozoites which infect red blood cells (erythrocytes). 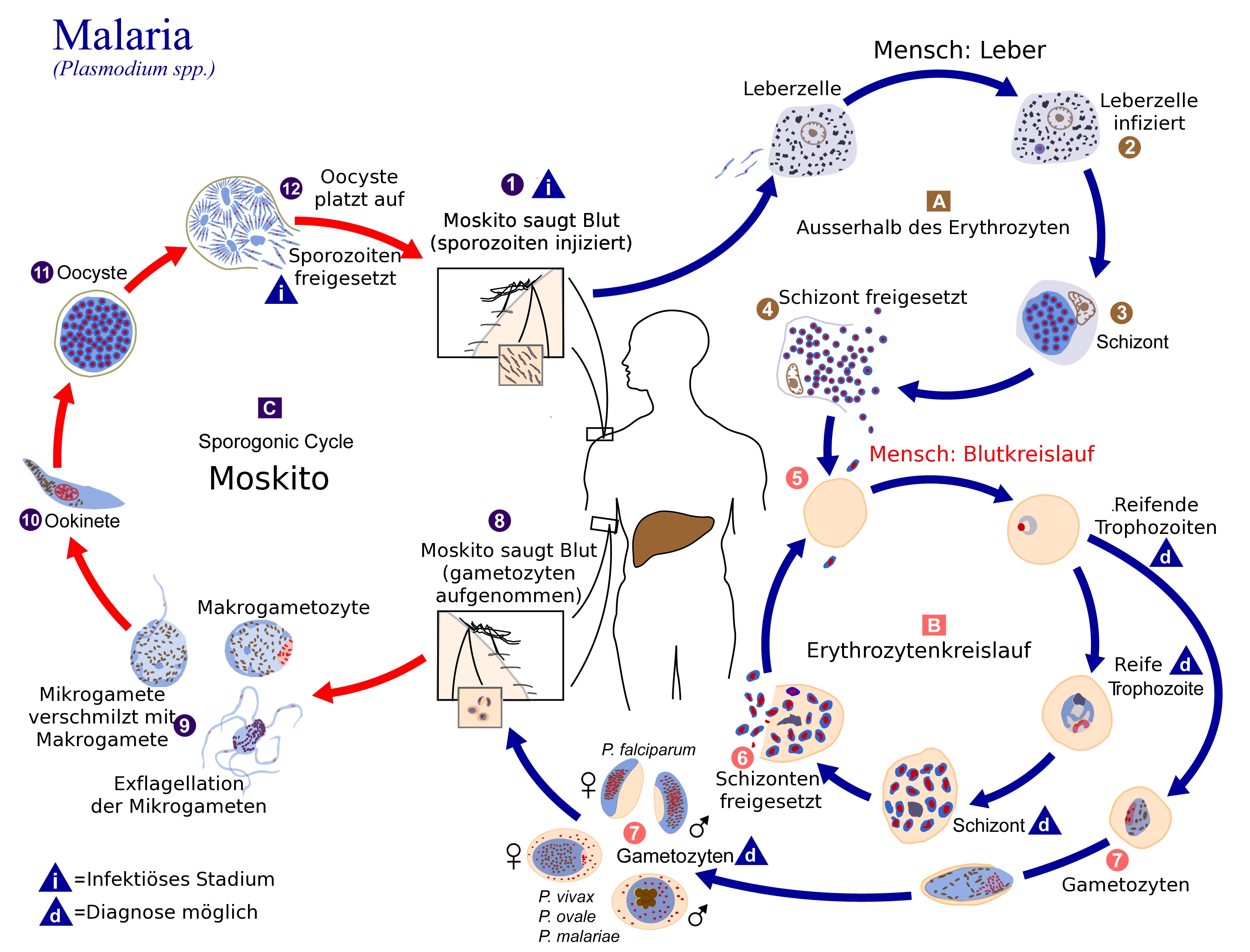
Plasmadium falciparum life cycle.
Graphic: Chb, Wikipedia.
There, the parasite reproduces asexually and develops into trophozoites in ring form. While some of these mature into schizonts, which in turn release merozoites, others develop into trophozoites, which transform into gametocytes (microgametes). These can only unite in the stomach of the mosquito and form zygotes. Blood from an infected person must therefore be sucked again. In the mosquito, the microgametes in turn become motile ookinetes, which migrate into the mosquitos' intestinal wall and form oocysts there. They again give rise to sporizoites, which are released by the mosquito sputum back into the human host during the next bite.(4)
To investigate these process, Hesperos, a human-on-a-chip company based in Orlando, has developed a 4-organ malaria disease model that contains human tissue constructs. The cell types used include hepatocytes, a white blood cell type from the spleen (splenocytes), endothelial cells, and recirculating red blood cells cultured in a microfluidic system. The disease can be simulated by an infection with the malaria pathogen Plasmodium falciparum. Chloroquine is a commonly used drug for the treatment and chemoprophylaxis of malaria.
The scientists observed that there is an interaction between the integrated organ constructs and that red blood cells can circulate in the system. The "organs" can also be infected with a sensitive or resistant malaria pathogen in the model, and a systemic administration of therapeutic agents such as chloroquine is also possible. In the sensitive strain, treatment with chloroquine reduced the number of pathogens; in the resistant strain, this reduction was only moderately noticeable. Impairment of human cell viability could be determined as a function of exposure to chloroquine concentration, but it was only significantly expressed in the resistant variant.
Original publication:
Michael J. Rupar; Trevor Sasserath; Ethan Smith; Brandon Comiter; Narasimhan Sriram; Christopher J. Long; Christopher W. McAleer & James J. Hickman (2023). Development of a human malaria-on-a-chip disease model for drug efficacy and off-target toxicity evaluation. Scientific Reports 13:10509. https://doi.org/10.1038/s41598-023-35694-4
Additional information:
(1) https://hesperosinc.com/new-publication-in-nature-scientific-reports-malaria-on-a-chip/
(2) https://tropeninstitut.de/aktuelle-krankheitsmeldungen/28.09.2021-afrika-malaria
(3) https://www.nejm.org/doi/full/10.1056/NEJMoa2101746
(4) https://www.msdmanuals.com/de-de/profi/multimedia/figure/lebenszyklus-von-plasmodium




 Dr. rer. nat.
Dr. rer. nat. Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.
Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.