Working Group – a Portrait: Aachen Centre of
Technology Transfer in Ophthalmology (ACTO)
Our newest article focuses on a special method developed by ACTO, the Ex Vivo Eye Irritation Test (EVEIT). Like the Bovine Corneal Opacity/ Permeability Assay1 (BCOP), the method is an animal-free alternative to the Draize test that uses living rabbits to test eye irritation.
ACTO is an affiliated institute of RWTH Aachen University, i.e. a scientific institute that is organisationally, financially and legally independent but is nonetheless associated with a university, meaning that innovation is less hampered by bureaucracy. The Aachen Centre of Technology Transfer in Ophthalmology was founded in 1998. It works at the interface between basic research and applied research. ACTO’s guiding principle is interdisciplinary scientific cooperation between physicians, basic researchers, technicians, biologists, computer scientists and engineers, as well as meshing basic research and applied research in ophthalmology. The research focuses are on the development of artificial cornea implants and animal-free tests for chemicals on the cornea. Additionally, new bases for the tolerance of eye drops and an online eyesight test are being developed. In order to be able to manage these tasks, ACTO runs research labs in both Cologne and Aachen.
Human eye with a cornea implant after one week.
Photo: ACTO
ACTO’s chairman is Prof. Dr. med. Dr. h.c. Norbert Schrage. He has been chief physician at the Ophthalmology Clinic in Cologne Merheim since 2004 and before that was acting director of the Ophthalmology Clinic at RWTH Aachen (1).
The vice-chairman is Univ.-Prof. Dr. med. Peter Walter, director of the Ophthalmology Clinic at RWTH Aachen. Prof. Schrage was recently awarded an honorary doctorate by the Pirogov Russian National Research Medical University “for research in the area of the development of corneal prostheses and research into the treatment of corrosive injuries of the eye” (2).
Ex Vivo Eye Irritation Test (EVEIT)
ACTO has developed a replacement method for the Draize test. The Draize test, which uses the eyes of living rabbits, is highly controversial, as it is painful and not very predictive. A principal criterion of the replacement method EVEIT is the ability to observe wound healing under exactly reproducible experimental conditions without testing on animals (3). The scientists successfully cultured the corneas of rabbits slaughtered for food in chambers for 28 days whilst keeping the cells’ metabolism stable. The test chemical is then applied to the cornea culture, after which damaged areas can be investigated using a variety of methods. Conversely, the healing process of an artificially damaged cornea culture can be investigated in vitro, for instance after a new pharmaceutical drug has been administered to the culture.
The test can also be used to test repeated substance applications with up to 100 applications per day.
Hornhaut in EVEIT-Kammer
Cornea in the EVEIT chamber.
Photo: ACTO.
The in vitro investigation of acute eye irritation uses four methods to test corneal opacity and changes in the superficial and deeper layers of the cornea – microscope, optical coherence tomography (OCT)2, biochemistry and histology – thus much more precisely than in tests on rabbits. In a project funded by the German Federal Ministry of Education and Research and in cooperation with the Institute of Semiconductor Electronics/Institut für Halbleitertechnik (IHT) at RWTH University Aachen and the company Innolabtec GmbH, it could be demonstrated that the EVEIT combined with OCT technology delivers results that can definitely match up to the results of animal experiments. In 2010, Dr. Spöler (IHT) was awarded the Science Award by the “European Partnership for Alternative Approaches to Animal Testing” (EPAA) for this development (1).

Monitoring corneal healing with four injured areas with the aid of fluorescein3 and blue light
Photos: ACTO
Optical Coherence Tomography
OCT uses light with a short coherence length to measure distances in light-scattering materials. The object under examination is scanned point by point. OCT is currently mainly used for examining the retina or back wall of the eye. The method allows light to penetrate quite deeply into tissue (up to 3 millimetres). Definition and measurement speed are comparatively high.
Coherence length is an optical term that describes a maximum difference in path lengths or time that two rays of light from the same source may have to create a spatially and temporally stable interference pattern when their paths cross. The interference is the alteration in amplitude as the sum of the magnitudes of two or more superposed waves.
A test substance in an in vitro test can cause the cornea being investigated to shrink. This loss of layer thickness is displayed by coherence tomography as an increased signal amplitude (4).
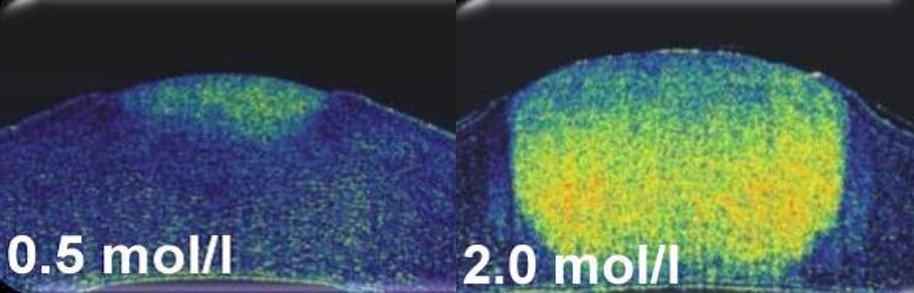
Depths of injury caused by caustic soda (NaOH) with the same application times and different concentrations, as determined by optical coherence tomography (OCT).
Photos: ACTO.
Experimental setup in an incubator.
Photo: ACTO.
Extremely painful and outdated: the Draize test
The American toxicologist John Henry Draize developed the test named after him using rabbits’ eyes in the 1940s, in order to test for possible irritation by substances such as cosmetics or industrial chemicals. The substance is applied to the cornea of a living rabbit and then observed over a period of days or even weeks to determine whether corneal cloudiness, corneal damage or conjunctivitis result. Nowadays the test on rabbits is conducted if in vitro alternative tests have already been conducted and the results did not demonstrate that the substance was potentially irritant. The OECD recommends conducting an integrated test strategy for the investigation of acute eye damage or irritation and only then considering an animal test when appropriate and necessary, i.e. when all other measures have been employed, such as weight-of-evidence4 analyses of data already available, validated and approved in vitro tests and all available alternative methods. For the Draize test, the substance is dripped into the animals’ conjunctiva (OECD Test Guideline no. 405 from October 2012, (5)).
The Draize test has been controversial for a long time and has widely been replaced. Regardless of the extreme stress for the animals – damaging the cornea and conjunctiva is extremely painful – there are also numerous differences between the corneas of rabbits and humans, whose safety the tests are generally meant to serve. Scientists speak of coincidental results, and each laboratory can arrive at different results.
Some examples of species differences are (6):
- rabbits’ eyes are ten times more permeable for water-soluble substances than the human eye
- rabbits’ pain threshold is higher than that of humans
- rabbits’ corneas are much thinner than humans’
- rabbits’ lacrimal apparatus (tear glands and ducts) is less effective than that of humans
- unlike rabbits, humans are able to form vacuoles in the corneal epithelium as a response to toxic substances
- the pH of rabbits’ tears is alkaline, whereas human tears tend to be pH-neutral
To be a thing of the past: rabbit in a Draize test
Photo: People for the Ethical Treatment of Animals (PETA)
As of September 2009, the OECD allows a variety of in vitro tests as replacement methods for verifying serious irritation potential, e.g. using isolated bovine corneas (from “slaughterhouse waste”) in the so-called BCOP Test, OECD Test Guideline 437, (7, 8). In addition to the BCOP Test there are further tests that can also be used as alternatives to the test on rabbits, e.g. the Isolated Chicken Eye Test5 (ICE, OECD Test Guideline 438) using the eyes of slaughtered chickens. There is also the Human Corneal Epithelium-Test6 (HCE) and the HET-CAM Test7 using incubated chicken eggs. The last two tests, however, have not been approved on a regulatory level. A number of scientists have specialised on the development of cornea models based on human cells (9, 10, 11, 12, 13), but none so far could completely replace the Draize test. For this reason it is nessessary to do further research and validation on replacement methods.
GHS Categories
The internationally agreed Globally Harmonized System of Classification and Labelling of Chemicals (GHS) defines the classification and labelling standards for chemicals, with substances denoted using one or more symbols and a signal word.
Chemicals can be assigned to one of three categories with regard to hazards to eyes:
Category 1: The substance can cause serious eye damage
Category 2A: The substance can cause serious eye irritation
Category 2B: The substance can cause eye irritation (14).
Can human cornea be produced artificially using stem cells?
Stem cell research has made considerable progress: With the help of tracer molecules, American scientists recently discovered so-called limbal stems cells in the human eye, with which they successfully cultured human cornea. The method is regarded as very promising for the future treatment of patients with eye injuries (15, 16, 17).
With continued technological advances, it is very probable that induced pluripotent stem cells will allow the development of complete corneas on a large scale for use as a replacement method for eye damage and irritation tests. For instance, the in vitro ocular model developed by Across Barriers (human cornea construct) comprises human corneal epithelial cells (HCE-T cell line) and a human corneal keratinocyte cell line (HCK-Ca cell line). The model is already used for testing the permeation and tolerability of ophthalmic drugs (9, 10, 11, 18).
“…much more sensitive and precise…”
InVitroJobs interviewed Prof. Dr. med. Dr. h.c. Norbert Schrage, founder and chairman of ACTO e. V.
InVitroJobs: Can you give us a brief description of your innovation?
Prof. Schrage: The long term goal of the Ex Vivo Eye Irritation Test (EVEIT) developed by ACTO is to replace the Draize test and animal experiments in pharmacological research into the eye. The eyes of slaughtered rabbits are treated so they can be cultured for several weeks. This provides a method that makes tests on living animals unnecessary and at the same time delivers results that are as meaningful or even much more meaningful. A central criterion of this method is the healing of wounds and chemical injuries in vitro. The tests conducted on the cornea serve the purpose of recognising efficacy, tolerability and side-effects – such as calcification caused by eye drops that contain phosphate – before administration to humans.
InVitroJobs: Which substances can be tested with the EVEIT?
Prof. Schrage: The EVEIT can be used for testing chemical, pharmacological and cosmetic substances or products.
InVitroJobs: What is the significance of this technology for replacing animal tests on rabbits?
Prof. Schrage: The purpose of the EVEIT system is to completely replace animal experiments with substances, chemicals or drugs applied to the eye. ACTO was able to prove that EVEIT is much more sensitive and precise than the Draize test, as the substance being tested can be applied in a much more controlled fashion, the effect observed and the substance rinsed off. One can distinguish mechanical and chemical damage, which is always a mixed result in animal experiments. The test is also much quicker and more exact than an animal experiment. Not only that, EVEIT can also prove delayed onset damage to the eye.
InVitroJobs: How many animals can be saved with this test?
Prof. Schrage: In 2007, tests were conducted on more than 2.6 million animals in Germany, with about 17,000 organ-specific tests for skin and eye irritation. These animal tests could be replaced by EVEIT.
InVitroJobs: What is the difference between your test and the Bovine Corneal Opacity/Permeability Assay (BCOP)?
Prof. Schrage: The BCOP can only test chemicals in the category CAT18 with regard to their acute effect, i.e. the change in transparency, measured using a light transmission method that is rather unusual for an ophthalmologist and hard to calibrate, and the change in permeability for sodium fluorescein. With the EVEIT system not only can these methods be used, but thanks to the culture we also see healing or lack thereof within a defined time period. This means that the rabbit corneas kept in the culture medium and not only treated with category CAT1 chemicals can answer the vital question as to how permanent damage is, i.e. will an injury heal, and if so will it scar or will there be a complete restoral of healthy tissue? This has been the central question for me as an eye specialist in the case of the 1,500 clinic patients with eye damage that I have seen in the course of my professional life. This is the decisive difference relevant to humans between the EVEIT as a clinically relevant test and the BCOP.
InVitroJobs: The crucial point is healing wounds in vitro: Can you give us an idea of what that looks like?
Prof. Schrage: For the long-term Ex Vivo Eye Irritation Test (EVEIT-longterm), the corneas of rabbits slaughtered for food production are removed and mounted in an artificial eye chamber, where they are nourished with a culture medium and monitored under a microscope so that a healthy cornea can stay alive in the culture for up to 28 days. This method is familiar from medical use of corneal cultures and transplantation of donated human corneas. The cornea’s condition is monitored using biochemical readings, inspection of clarity, epithelial (surface layer) stability and endothelial (inner layer) function. On day 2, once these criteria have been fulfilled, the corneas used in healing tests are injured in four defined areas, to which drops of the substance to be tested are then applied. The corneas are cultivated for between three and twenty days in an incubator whilst continually supplied with culture medium. Each day, photos are taken of the corneas and the individual injured areas using fluorescence photography9 and with the help of optical coherence tomography (OCT), and the vital functions of the cornea are observed by determining glucose and lactate. The daily observation allows us to measure and see exactly if and how fast the injured areas of the cornea heal. This is evaluated digitally and documented in a healing curve for each cornea.
InVitroJobs: The commonly used BCOP is normally combined with further tests, such as CAMVA10 or HET-CAM. Is that also necessary with your test?
Prof. Schrage: Additional tests such as CAMVA or HET-CAM are not necessary within the framework of the questions being investigated using the EVEIT, as these deliver answers to questions regarding broader toxicity, which are rather unspecific with regard to the eye. The haemorrhages in HET-CAM correspond to OCT changes in the cornea. As supplementary parameters we have histological stains, tests of permeability and proof of corneal vitality by quantification of glucose and lactate. Especially direct measurements of opacity11 and permeability are possible with the living corneal model. We also use the much more precisely differentiating optical coherence tomography to investigate the cornea.
InVitroJobs: How is the endpoint12 measured/detected? How are the endpoints for light, medium and serious eye damage, and light, medium and serious eye irritation classified? Do you have a measuring device you just need to read a value from?
Prof. Schrage: Normally it’s the complete healing process of the negative control run with each test series that defines the endpoint. As a rule three test days are sufficient. A longer observation period for more specific issues is possible. A certain exposure classification is possible, for instance by varying the application frequency (e.g. hourly or daily application of the test substance).
The endpoints are defined as: epithelial healing in fluorescein test3, corneal opacity in backlighting, corneal opacity in optical coherence tomography including penetration depth of the lesion in OCT, global permeability of the cornea, and glucose and lactate measurement in the cultured cornea. Histological endpoints and dynamic observations are clinically possible due to the longer test period between three and six days.
InVitroJobs: According to our information, the test on rabbits is still used in Europe when the eye irritation test delivers a negative result, in order to test whether mild irritation can still result that was not detected by the BCOP and the other tests. How does that relate to your test?
Prof. Schrage: In our opinion, our test can determine the irritation threshold very exactly, as we can administer the drops repeatedly and not just once as in previous methods. Thus we were able to show that substances that tested as completely harmless in the one-off test actually did disturb epithelial healing or were even toxic when the application frequency was increased. A good example is our publications on classic preservatives in ophthalmology, such as benzalkonium chloride or Purite®.
Quote: The Ex Vivo Eye Irritation Test (EVEIT) in evaluation of artificial tears: Purite-preserved versus unpreserved eye drops. Schrage N, Frentz M, Spoeler F. Graefes Arch Clin Exp Ophthalmol. 2012 Sep;250(9):1333-40. doi: 10.1007/s00417-012-1999-3. Epub 2012 May 15.
The one-off application did not show any toxicity, however in the case of hourly application a considerable disturbance of epithelial healing or irreversible degeneration were detectable after three days. This corresponds to clinical results in patients who react to frequently administered drugs that contain preservatives with toxic side-effects. This means that the EVEIT system can help to titrate (adjust) the maximum dose. That enables an extremely exact differentiation of the term “toxic” in a way that can never be achieved in an animal experiment but typically results in humans. Paracelsus’ adage that the dose makes the poison was never more apt than here. It’s senseless to administer a highly concentrated substance to the eye and think we can measure toxicity that way. This can be illustrated with a simple example that I do not recommend as a self-experiment: Strew a few grains of table salt into the conjunctiva of the eye and describe the effect of table salt. You will notice how irritant and toxic this foodstuff is when applied this way. Therefore, the threshold values for one-off and repeated application must be defined. EVEIT is especially suitable for this. The test has already been used to test several tear-replacement solutions and new drugs that were approved by the responsible departments of the Federal Institute for Drugs and Medical Devices (BfArM13).
Quote: Semifluorinated alkanes as a liquid drug carrier system for topical ocular drug delivery. Dutescu RM, Panfil C, Merkel OM, Schrage N. Eur J Pharm Biopharm. 2014 May 15. pii: S0939-6411(14)00158-1. doi: 10.1016/j.ejpb.2014.05.009
InVitroJobs: What is the difference between human and animal corneas?
Prof. Schrage: Rabbit corneas are the corneas of young animals, thus comparable to children’s corneas and not those of adult humans. These corneas are much more permeable and thinner and react more extremely than the adult human cornea does. That means that these tests are on the safe side in terms of risk assessment due to their higher sensitivity, and if anything too sensitive.
InVitroJobs: What are the chances of using your method as a high-throughput test?
Prof. Schrage: We are currently developing modifications to our systems, in order to address obstacles in the culture technology and flaws, and to unify and especially simplify the high demands on handling and application. There is a proven SOP14 that can be modified. We want our method to be brought to prevalidation (19) by the European Union (ECVAM15) and at the same time, without altering the culture conditions, establish a high- throughput method and validate it together with other partners in inter-lab tests. As hundreds of thousands of rabbits are commercially bred for slaughter in Germany, and breeding conditions and genetic uniformity are extremely standardised, we are not worried about running out of rabbits’ eyes.
InVitroJobs: Could one, for ethical reasons, at a later time use human corneas developed from a cell culture?
Prof. Schrage: Yes, if they were donated corneas not suitable for transplantation, one could also use human corneas in our test. This requires the consent of the donors themselves or their relatives. The EVEIT chamber is currently being further developed to that end, as rabbit corneas are somewhat smaller than human corneas. As culturing of the corneas also needs to be started immediately, there are considerable pharmaceutical legal requirements that need to be fulfilled, with current legislation making such a development very difficult. Part of the problem is also the fact that we don’t always immediately know whether a cornea is suitable for transplantation or not. The unused human donor cornea is altered by the culture process (in preparation for transplantation) in a way that makes an EVEIT culture no longer feasible. The culturing method would have to be adapted, something that a small institute cannot implement without external help in the form of generous donations.
InVitroJobs: What test phase is your test currently in?
Prof. Schrage: We are currently looking for funding and cooperation partners, in order to validate the test. The institute unfortunately cannot do this with its own resources. However, the prospects of receiving funding are good and we are very optimistic that we will obtain the validation within the next five years.
InVitroJobs: How has the industry reacted to the test?
Prof. Schrage: The industry is very interested in our test and there has already been funding by the Colipa (association of European cosmetic producers) and the EPAA (European Producer Associations Alliance). We would certainly like to involve the industry on the path to validation. The pharmaceutical industry is especially interested in the test, as it offers a simple method of collecting valid data on effects and side-effects of galenics and the pharmacokinetics of isolated corneas by conducting one-off tests, repeated tests and saturation tests on a single cornea.
InVitroJobs: Do you train students in the development and application of the test or replacement methods for animal tests?
Prof. Schrage: No, not at present. In the past, eight doctoral students have learned to use this test and worked independently with it.
InVitroJobs: Thank you for speaking with us.
Glossary:
1 Bovine Corneal Opacity/Permeability Assay: In this test corneas from slaughtered cattle are mounted in an apparatus. The opacity and permeability of the cornea a measured with a so-called opacitometer and a spectrophotometer using visible light (ICCVAM 2006 in EURL ECVAM DB-ALM: Protocol No. 127 (20,21).
2 Optical coherence tomograph: similar to ultrasound (see box above for description).
3 Fluorescein: a yellow-red xanthene compound that fluoresces green in alkaline solutions.
4 Weight-of-evidence analysis: A method for assessing strengths and weaknesses of available information (22).
5 Isolated Chicken Eye Test (ICE): Test for category 1 substances. Uses the eyes of chickens from slaughterhouses. The eyes are enucleated and mounted in an eye holder with the cornea positioned horizontally. The test substance and negative/positive controls are applied to the cornea. Toxic effects to the cornea are measured by a qualitative assessment of opacity, a qualitative assessment of damage to epithelium based on fluorescein retention, a quantitative measurement of increased thickness (swelling), and a qualitative evaluation of macroscopic morphological damage to the surface. (23).
6 Human Corneal Epithelium-Test (HCE): Reconstituted Human Corneal Epithelium. A three-dimensional epithelium model using human corneal cells for the eye irritation test (24).
7 HET-CAM Test: Hens Egg Test Chorio Allantoic Membrane, Alternative to the Draize test von 1985. The test substance is applied to the chorioallantoic membrane of an incubated chicken egg and the reaction to the substance studied (haemorrhage, vessel lysis and coagulation (25)
8 Category CAT 1: Global Harmonized System (GHS) hazard category, also see box above. Category 1: The substance can cause serious eye damage.
9 Fluorescence angiography: A harmless fluorescent dye is injected into a vein of the forearm and is transported to the retinal and choroid vessels in the eye, allowing photographic images to be taken of the fundus (eyeground) using a special camera (26).
10 CAMVA: Chorioallantoic Membrane Vascular Assay, an alternative test using incubated chicken eggs (27)
11 Opacity: cloudiness.
12 Endpoint: the target outcome of a trial.
13 BfArM: Federal Institute of Drugs and Medical Devices.
14 SOP: Standard Operation Procedure.
15 ECVAM (EURL ECVAM): European Union Reference Laboratory for alternatives to animal testing of the European Centre for the Validation of Alternative Methods (28).
Bibliography and internet sources:
(1) http://www.rwth-aachen.de/go/id/bqbt/?#aaaaaaaaaaabqbu
(2) http://idw-online.de/pages/de/news592629
(3) http://www.acto.de/pages/de/projekte/eveit.php#Testprinzip
(4) Spöler, F, Frentz, M, Först, M, Kurz, H & Schrage, NF (2008): Analysis of hydrofluoric acid penetration and
decontamination of the eye by means of time-resolved optical coherence tomography. Burns 34: 549-555.
(5) http://www.oecd-ilibrary.org/docserver/download/9712201e.pdf?expires=1405453242&id=id&accname=guest&checksum=8B94846B1CDF0F314ACE10865AA83498
(6) https://web.archive.org/web/20070814145631/http://www.curedisease.com/Perspectives/vol_1_1989/Problems%20with%20the%20Draize.html
(7) http://www.invitrojobs.de/index.php/de/aktuelles-archiv/72-in-vitrobcop-testalsersatzfuertestsankaninchenaugen.html
(8) http://www.invitrojobs.de/index.php/de/aktuelles-archiv/185-arbeitsgruppe-im-portrait-kurzzeit-toxikologie-der-abteilung-experimentelle-toxikologie-und-oekologie-basf.html
(9) http://www.invitrojobs.com/index.php?option=com_content&view=article&id=327:arbeitsgruppe-im-portrait-across-barriers&catid=2:news&Itemid=29
(10) Bock, U., Haltner, E., Guzman, G. & Reichl, S. (2012): Characterisation of Human Cornea Constructs for the Rapid Screening of Preservatives and Evaluation of Their Use in Permeability Assays to Reduce Animal Testing. Abstract for the European Congress on Alternatives to Animal Testing, Linz.
(11) Guzman, G., Reichl, S., Bock, U. & Haltner, E. (2012): Optimisation of Human Cornea Constructs For a Rapid Characterisation of Pharmaceutical Preparations, Excipients and Chemical Compounds. Abstract for the European Congress on Alternatives to Animal Testing, Linz.
(12) Reichl, S. & Müller-Goymann, C. C. (2004): Künstliche Hornhaut als In-vitro-Modell. http://www.pharmazeutische-zeitung.de/index.php?id=titel_20_2004
(13) Reichl, S., Bednarz, J., C C Müller-Goymann, C. C. (2004): Human corneal equivalent as cell culture model for in vitro drug permeation studies. Br J Ophthalmol. 88: 560–565.
(14) Globally Harmonized System (GHS) of Classification and Labelling of Chemicals Produced by SCHC-OSHA Alliance
GHS Information Sheet Workgroup. Info Sheet #5 Eye Damage / Eye Irritation May, 2010.
(15) http://www.wallstreet-online.de/nachricht/6850030-limbale-stammzellen-hornhaut-nachwachsen
(16) http://www.masseyeandear.org/news/press_releases/recent/2014_Nature_Growing_Corneas/
(17) Ksander, BR, Kolovou, PE, Wilson, BJ, Saab, KR, Guo, Q, Ma, J, McGuire, SP, Gregory, MS, Vincent, WJB, Perez, VL, Cruz-Guilloty, F, Kao, WWY, Call, MK, Tucker, BA, Zhan, Q, Murphy, GF, Lathrop, KL, Alt, C, Mortensen, LJ, Lin, CP, Zieske, JD, Frank, MH, & Frank, NY (2014): ABCB5 is a limbal stem cell gene required for corneal development and repair. Nature Letter. doi:10.1038/nature13426. http://www.nature.com/nature/journal/vaop/ncurrent/full/nature13426.html
(18) http://www.acrossbarriers.de/services/in-vitro-pharmakokinetik/okular/
(19) Adriaens, E, Barroso, J, Eskes, C, Hoffmann, S, McNamee, P, Alépée, N, Bessou-Touya, S, De Smedt, A, De Wever, B, Pfannenbecker, U, Tailhardat, M & Zuang, V (2014): Retrospective analysis of the Draize test for serious eye damage/eye irritation: importance of understanding the in vivo endpoints under UN GHS/EU CLP for the development and evaluation of in vitro test methods. Arch Toxicol 88: 701–723.
(20) EURL ECVAM DB-ALM: Protocols: Bovine Corneal Opacity and Permeability (BCOP) Assay. DB-ALM Protocol No. 127. http://ecvam-dbalm.jrc.ec.europa.eu/public_view_doc2.cfm?id=9A506ACE702B2EFB9471CBE1CBAA1945579489581FB95A26F52B280413D1D53EA3291B895581F634
(21) http://www.invitrojobs.de/index.php/de/aktuelles-archiv/72-in-vitrobcop-testalsersatzfuertestsankaninchenaugen.html
(22) http://echa.europa.eu/documents/10162/13655/pg_report_weight_of_evidence_en.pdf
(23) http://www.oecd-ilibrary.org/environment/test-no-438-isolated-chicken-eye-test-method-for-identifying-ocular-corrosives-and-severe-irritants_9789264076310-en
(24) Doucet, O, Lanvin, M, Thillou, C, Linossier, C, Pupat, C, Merlin, B & Zastrow, L (2006): Reconstituted human corneal epithelium: A new alternative to the Draize eye test for the assessment of the eye irritation potential of chemicals and cosmetic products. Toxicology in Vitro 20/4: 499–512.
(25) http://pharmazie-lehrbuch.de/kapitel/4-3.pdf
(26) http://www.vincentius-kliniken.de/kliniken-institute/augenklinik/schwerpunkte/fluoreszenzangiographie.html
(27) http://www.mbresearch.com/camva.htm
(28) http://ihcp.jrc.ec.europa.eu/our_labs/eurl-ecvam
Further Reading:
(29) Alépée, N, Bessou-Touya, S, Cotovio, J, de Smedt, A, de Wever, B, Faller, C, Jones, P, Le Varlet, B, Marrec-Fairley, M, Pfannenbecker, U, Tailhardat, M, van Goethem, F & McNamee, P. (2013): Cosmetics Europe multi-laboratory pre-validation of the SkinEthic™ reconstituted human corneal epithelium test method for the prediction of eye irritation. Toxicol In Vitro 27(5): 1476-1488.
(30) H. Thomason & D. J. S. Montagnes (2013): Developing a quick and inexpensive in vitro (non-animal) bioassay for mascara irritation. International Journal of Cosmetic Science, 2013, 1–6. doi: 10.1111/ics.12106
(31) Scott L, Eskes C, Hoffmann S, Adriaens E, Alepee N, Bufo M, Clothier R, Facchini D, Faller C, Guest R, Harbell J, Hartung T, Kamp H, Varlet BL, Meloni M, McNamee P, Osborne R, Pape W, Pfannenbecker U, Prinsen M, Seaman C, Spielmann H, Stokes W, Trouba K, Berghe CV, Goethem FV, Vassallo M, Vinardell P, Zuang V (2010) A proposed eye irritation testing strategy to reduce and replace in vivo studies using Bottom-Up and Top-Down approaches. Toxicol In Vitro 24: 1-9




 Dr. rer. nat.
Dr. rer. nat. Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.
Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.