Siblings.
Photo: Sathyatripodi, Pixabay.
About 20 years ago, scientists tried to find new solutions for reproduction toxicology without animal suffering. For example, between 2004 and 2009, the EU project ReProTect was carried out in a more comprehensive manner. In this project, 14 methods for testing on reproduction and development toxicology were developed and evaluated in a test battery.
In the meantime, technological progress and scientific knowledge have developed further. Reproductive organs can be cultivated on miniaturised multi-organ chips, which are even able to simulate the hormone cycle. In the future, induced pluripotent stem cells will offer the potential to cultivate the desired human tissue and, if necessary, to shape it using 3D printers. Genetic modification allows the incorporation of reporter genes into cells, which can, for example, be used to quickly detect a damaging or disruptive effect with a fluorescence signal, which can also be detected in high throughput. Here we present just a few highlights.
Replacement of Animal Experiments in Reproduction Toxicology: Small Steps
Between 2004 and 2009, an EU project was carried out with the aim of finding new approaches to reproduction toxicity by combining in vitro methods with tissue and sensor technologies (1, 2). 32 European research groups from academia, industry, small and medium-sized enterprises and government institutions were involved. A number of alternative test systems were developed which had to cover the different stages of the reproductive cycle. The methods were evaluated by an external, independent expert group in a ring feasibility study with a fixed number of test chemicals.
As part of this EU project, six receptor binding tests have been developed to investigate hormone-reactive substances, of which the so-called AR CALUX® has finally been incorporated into the OECD test guidelines. It is a reporter gene assay used to test the androgenic and anti-androgenic activity of compounds in vitro. Androgen-active substances bind to the so-called androgen receptor and trigger an effect characteristic of the androgenic effect. Anti-androgens, on the other hand, block the androgen receptor and thus prevent the androgen effect.
A similar assay was constructed with a human osteosarcoma cell line (U2-OS) incorporating a human estrogen receptor (3) to measure the estrogenic and anti-estrogenic potential of chemicals. The assay was validated by the OECD in 2015 and was considered suitable for inclusion in the OECD "Performance Based Test Guideline for Stably Transfected Transactivation In vitro Assays to Detect Estrogen Receptor Agonists and Antagonists" (TG 455), as part of a tiered testing strategy (4), in Appendix 4. Other receptor binding assays were the MELN assay with a breast cancer cell line (MCF-7) incorporating an estrogen responsive gene (5) and the MTS assay with a human prostate adenocarcinoma cell line (LNCaP) to identify molecules targeting prostate function (6).
Five other tests provided information on fertility disorders (folliculogenesis and oogenesis), an in vitro fertilisation test on fertility disorders and a peri implantation test with a mouse zygote, whose development was followed up to the blastocyst stage. The Ishikawa cell test provided information on implantation and estrogenic activity (7). A modified comet assay (ReProComet) was used to detect DNA strand breaks in bovine sperm. Since the binding of the substance to the DNA of sperm normally occurs only after fertilisation and cannot be detected in sperm itself, as these have no repair enzyme system, an extract of human breast cancer cells (HeLa) was added to the bovine sperm embedded in agarose for the test (8, 9).
To study the placental patency of bisphenol A, an estrogen-like substance (10), scientists combined a trophoblast cell line (trophoblast is the embryo's nourishing envelope cell layer) and human placental explant cultures to perform placental perfusion tests. The combined tissue was able to produce the important pregnancy hormone hCG. Due to the hormone-active test chemicals, the hormone amount of hCG and the expression of caspase 3, an apoptosis protein, increased in the placenta explants. Trophoblast cells were damaged (11). To test the placental permeability of substances, researchers constructed a placental perfusion model with two independent circuits corresponding to the blood circulation of the mother and the foetus (12, 13, 14).
In tests for embryonic development, three models were favoured respectively developed: One was the Whole Embryo Culture (WEC) with rat embryos, which investigated growth and developmental damage to the embryo. It was established in chemical testing. An other was the murine embryonic stem cell test (mEST), which was used to investigate the differentiation inhibition of mouse embryonic stem cells into heart cells. The third test, the ReProGlo Assay, was investigating the inhibition of the so-called Wnt signalling pathway (more precisely: canonical Wnt/catenin signalling pathway) by drugs. Wnt is one of six important signalling pathways and is active during the regulation of early embryogenesis. Disturbance of this pathway leads to abnormal embryonic development. In some cases, the test required the previous metabolic activation by liver cells (15, 16). Reviewees, however, criticized the low predictive accuracy of this test (17).
Researchers tested all these developments in the form of an comprehensive test battery with ten test substances and evaluated the methods developed to replace animal experiments as " carefully positive". They were able to identify the toxicologically relevant properties of the test substances with relatively high accuracy in the majority of cases. Only one reliable specific test for male fertility was missing (18). At the end, however, only a few developments were implemented in the test guidelines.
![]()
Fertility statue.
Photo: Domaris, Pixelio.de
In ReProTect, scientists also combined four already available software tools (CAESAR (Q)SAR model, the Derek for Windows expert system, the estrogen-receptor-binding profiler of the OECD/(Q)SAR Application Toolbox and a read-across model) in a weight-of-evidence approach to predict reproduction and developmental toxicity. However, due to the complexity of reproductive and developmental toxicity the results were not sufficient (19).
Current developments
Many scientists are convinced that, for instance, new chip systems with human tissue or imitated organs in the field of reproduction toxicity can provide information and make new therapeutic approaches possible. In recent years, developments in this field have progressed rapidly. Only a few procedures can be presented here.
In vitro fallopian tube model
A research group led by Dr. Jennifer Schön from the Leibniz Institute for Farm Animal Biology in Dummerstorf has succeeded in establishing a 3D cell culture model of the fallopian tube. However, the fallopian tubes here come from cattle and pigs. Embryos could even develop in the "fallopian fluid" of the model, so that early interactions between embryo and "mother" can be investigated with the model. Early embryonic losses are one of the most common reproductive disorders in humans and animals (20).
In vitro model of follicular growth
An American research team led by Prof. Theresa Woodruff from the Women's Health Research Institute at Northwestern University Feinberg School of Medicine in Evanston, Illinois, has succeeded in isolating, cultivating and further developing primary human follicles - the precursor of the female egg. Early stages of follicular development in vitro required their encapsulation in alginate and support by natural ovarian cortex tissue (21).
Male gonads in vitro
A Danish-Scottish research team led by Prof. Anders Juul and Dr. Ewa Rajpert-De Meyts from the Rigshospitalet of the University of Copenhagen has isolated human male fetal gonads from tissue of fetuses from abortions and cultivated them in a hanging drop culture. In this model retinoic acid was tested, which can cause severe malformations and therefore has an influence on embryonic development. A surplus of retinoic acid at the time of testicular development leads to a disturbance of germ cell development (22). Unfortunately, the cells originated from human embryos and for ethical reasons do not represent a real perspective in this form.
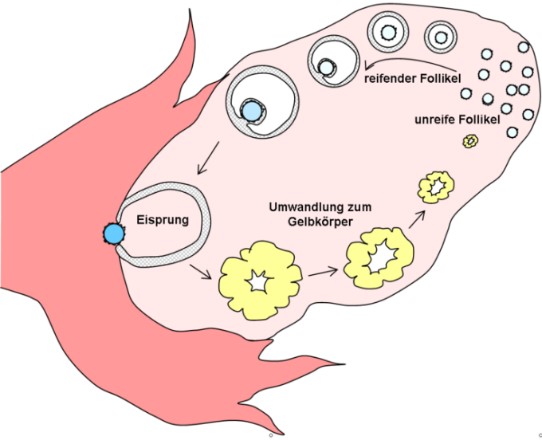
Oocyte maturation cycle in the ovary.
Graphic. H. Hoffmeister, Wikipedia.
Organ-on-a-chip developments
Conventional models derived from human cells look at toxicological processes isolated and distant from a normal physiological microenvironment. Therefore, scientists try to simulate microphysiology using organ-on-a-chip or human-on-a-chip models. Human tissue is integrated into a so-called microfluidic system, which is designed to mimic the complexity of human in vivo physiology in vitro. However, the simulation of the female and male reproductive tract, with several organs each and the complex hormone system, is currently still a particular challenge.
3D model of the human placenta
Scientists led by Prof. Aleksandr Ovsianikov from the Institute of Materials Science and Technology in Vienna have developed an artificial placenta model. A human trophoblast cell line (BeWo b30) and human primary placental villi endothelial cells were used for the model. Using a laser-guided 3D printing process, hydrogels can be used to produce high-precision shapes that can then be seeded with placenta cells. The organoid produced in this way grows on a microfluidic chip on which important biological parameters such as pressure, temperature or nutrient as well as drug delivery can be adjusted. The model can be used to form a placenta barrier with permeability for large proteins similar to natural tissue. Two separate chambers simulate the fluid flows in the fetal and maternal "organism". The cells are able to produce certain structural proteins (E-cadherin) and protein transporters (23).
Researchers from the Swiss Federal Laboratories for Materials Science and Technology (EMPA) under the direction of Dr. Tina Buerki-Thurnherr have also developed a placenta organoid model on the chip. They are investigating whether nanoparticles can cross the placental barrier (24).
Chinese researchers led by Prof. Jianhua Qin from the Dalian Institute of Chemical Physics (DICP) at the Chinese Academy of Sciences in Beijing have developed a disease model: They added bacteria to an in vitro constructed placental barrier in order to investigate inflammatory processes that can lead to malformations of the fetus or miscarriage during pregnancy (25).
Prof. Katharina Ribbeck from the Massachusetts Institute of Technology (MIT) in Boston and her team are working on similar research. They have developed a cervical mucosa model on the chip (26). This model is used to investigate the transport of bacteria through the mucous membrane. The rise of bacteria in the uterus during pregnancy can lead to premature birth.
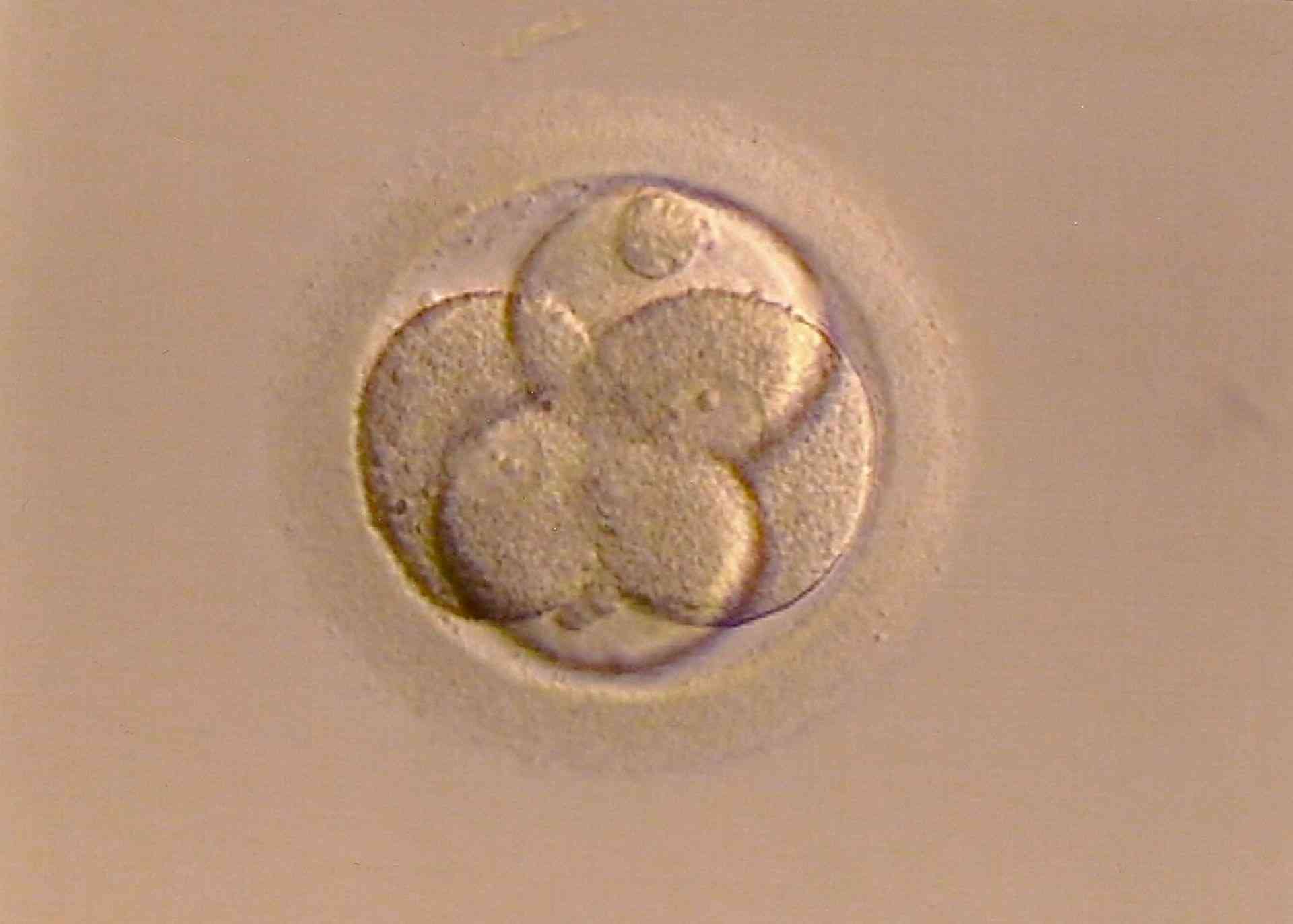
Development of a zygote (8-cell stage).
Photo: Bellezza87, Pixabay.
Simulation of the human female menstrual cycle ("Repro-on-a-Chip")
Under the direction of Prof. Theresa Woodruff, Director of the Women's Health Research Institute at Northwestern University Feinberg School of Medicine in Evanston, Illinois, a research team succeeded in reproducing the hormone profile of the human 28-day menstrual cycle in women. Several microfluidic systems and ovarian follicles of the mouse were used. Healthy ovarian tissue of female test subjects was not available and cannot be obtained for ethical reasons. One-, two- and multi-stage microfluidic platforms such as the EVATAR (27) formed a circuit between the cultured tissues of the female reproductive tract (ovary, fallopian tubes, etc.), The researchers were able to simulate follicle growth, follicle maturation and ovulation. During the follicular maturation process the tissue produced the hormones progesterone and 17b-estradiol. Single-, two- and multi-stage microfluidic platforms such as EVATAR (27) formed a circuit between the cultivated tissues of the female reproductive tract (ovarian, fallopian, cervical and liver tissues as well as uterine mucosal cells). They supplied the organ-like systems with the necessary hormones such as follicle-stimulating hormone (FSH), luteinising hormone (LH) and human chorionic gonadotropin (hCG) in a temporally coordinated regime using micropumps. Follicular growth, follicular maturation and an ovulation could be simulated. The tissue produced the hormones progesterone and 17b-estradiol during the follicular maturation process. With the exception of the ovaries and the follicles, all tissue came from human biopsies. It could be cultivated over a period of 28 days (28). The team is currently working on cultivating the various organ cultures on a single platform and thus establishing a complete female reproductive tract on a microfluidic platform. Then, the ovarian follicle hormones will flow via the cell culture media into the fallopian tubes, the uterus and the inner and outer layers of the cervix (endocervix and ectocervix). The system is currently being validated, the use of alternative cell sources such as induced pluripotent stem cells is intended (29).
It is impressive and groundbreaking that the American team of scientists has succeeded in simulating hormone regulation on the chip. This enables the researchers to investigate not only hormonal effects on reproduction, but also on other organ-like systems on the chip, such as the heart (30). The team is currently developing the DudeKUBE, an in vitro model that simulates the male gonads on the chip (31). The aim is to reproduce the male reproductive tract (DudeKUBE) on the chip in the same way as its female counterpart EVATAR (32).
Animal-free methods for developmental neurotoxicity (DNT)
During pregnancy, the development of nerve cells in the fetus may be disrupted, leading to altered nerve cell connections. Environmental toxins can inhibit the division, differentiation or migration of nerve cells (33). These disorders often manifest later in learning disabilities, developmental delays, autism spectrum disorders as well as attention deficit and hyperactivity disorders. With regard to the causes, both environmental chemicals and drugs are discussed. It is estimated that about 4% of prescription drugs have been withdrawn from the market due to observed adverse neurological effects (34).
These disorders are investigated with developmental neurotoxicity tests (DNT tests) in rats. So far, these tests have not been required as standard by the regulatory authorities. However, according to the European Chemicals Directive REACH, they are additionally carried out if there has been evidence of DNT in the previously conducted extended one-generation study in rats. This is the case if disturbances in cholinesterase (liver damage) or changes in thyroid hormones or the estrogen balance have been found.
Scientists such as Prof. Marcel Leist, who investigates DNT himself, criticize, that most DNT endpoints in animals cannot be measured at all, e.g. speech disorders, an impairment of attention duration or the IQ (35). Studies have found significant species differences in DNT reactions between rodents and humans with in vitro test systems (36).
A research team led by Prof. Ellen Fritsche from the Institute for Environmental Medicine Research in Düsseldorf used so-called neurospheres from induced pluripotent stem cells (hiPSCs) of humans, mice and rats to investigate the influence of environmental chemicals on early brain development. Cell proliferation, cell differentiation, cell migration and apoptosis - all processes that take place in the developing brain - were tested (37).
A possibly suitable test is the NeuriTox test from the research group of Prof. Marcel Leist, head of the Doerenkamp Zbinden Chair at the University of Constance. It is based on a cell line of human immortalised neuronal cells of the midbrain, the so-called LUHMES cells. In order to assess the suitability for tests, a large number of substances have already been screened (38).
Dr. Stefan Schildknecht from the University of Constance inserted a fluorescence gene into the mitochondria of the In a subtype of this cell line to monitor the movement of mitochondria, a possibility to assess the degeneration of nerve cells caused by test substances (39). The test has been refined that it can be used in high-throughput procedures (40).
Regulatory authorities such as EFSA are working together with scientists on a cost-effective testing strategy based on a reliable in vitro test battery in order to identify DNT hazards and initiate measures to reduce exposure to these chemicals (37). In so doing, they want to use the so-called Adverse Outcome Pathway (AOP) concept. AOPs describe processes that originate from a triggering molecular event on the cell surface and lead to a damaging effect via a chemical stress factor and a cascade of biological reactions upwards through different biological levels (organelles, cells, tissues, organs, entire organisms) over time. If one of these AOPs is demonstrably affected, this would allow conclusions to be drawn about the DNT properties of the substance.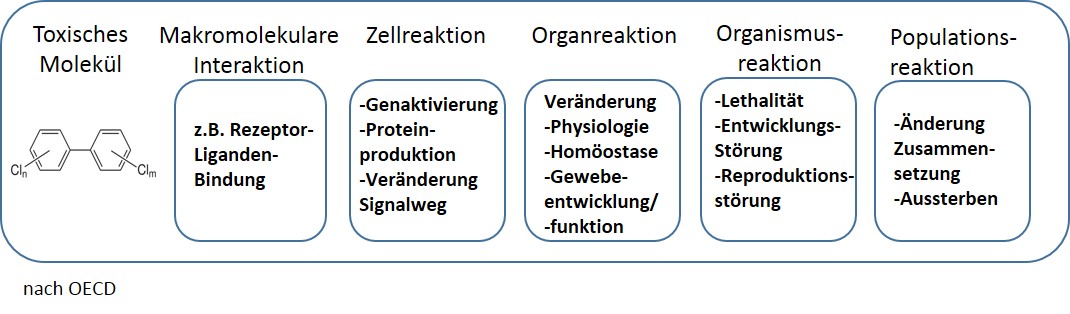
Animal testing and non-animal testing procedures in chemicals legislation
Chemicals produced in quantities of 10 tonnes or more per year (REACH Annex VIII and IX), pesticides, food additives, medicines and other substances must be tested for their potential to cause reproductive or developmental harm.
Reproductive toxicity tests evaluate the tendency of a substance to cause damage to the reproductive system. A substance is toxic for reproduction if it impairs male or female fertility, impairs development, or causes non-heritable damage to offspring (41). Developmental toxicity tests investigate the extent to which exposure may be harmful to a developing embryo or foetus in the womb. In general, harmful effects can occur at any stage of development, leading to death, deformation, altering growth or functional deficits.
Reproductive and developmental toxicity tests are usually associated with the use of a large number of animals. Due to the European agreement on the reduction of animal testing, scientists and regulatory authorities have tried to reduce the consumption of laboratory animals by new guidelines, e.g. to allow an extended one-generation study instead of a two-generation reproductive study. However, the acceptance of alternative methods to animal testing looks modest: Only the in vitro estrogen and androgen receptor tests are in the library of test guidelines (TG 455, 458 and 493).
Prenatal developmental toxicity study (TG 414)
The prenatal developmental toxicity study is intended to provide general information on the effects of prenatal exposure of the developing child organism (death, structural abnormalities or growth changes of the foetus) using pregnant laboratory animals. Functional deficiencies as an important part of development are not addressed by this Directive. Test species are the rat and - as a non-rodent - the rabbit. Here at least 260 animals are consumed per substance and test species (42).
Extended one-generation study (TG 443)
In the extended one-generation study, viability, health status of newborns, developmental stage at birth, physical and functional development to adult stage, gonadal function, oestrus cycle, epididymal sperm maturation, maturity, conception, pregnancy, birth process and lactation are investigated. The test substance is administered to the animals via drinking water, a gavage tube, inhalative or dermal, depending on the substance characteristics (43). At least 680-1,000 animals are consumed per substance and test species. (For comparison: in the two-generation study there are about 3,200 (44).
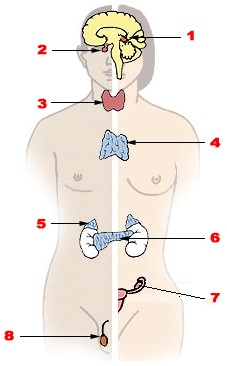
Reproduction: a complicated interplay between organs and hormonesThe reproductive system consists of several organs composed of different tissue and cell types. They produce various factors and hormones. Hormones not only control the organs and tissues of the reproductive tract, the production of germ cells and determine fertility, but they also communicate with organs and tissues of different systems throughout the body. There are gender-specific differences in disease development and reaction to drugs (30).Overview of the glands that secrete hormones via the blood system (endocrine system): |
Drug development
Two guidelines, the ICH guidelines S5(R2) and S5(R3), provide information on animal experiments to be carried out. The S5(R3) examines the female and male reproductive function over three generations in several steps. According to the regulations, studies should be carried out on mammalian species (especially rats). Only in embryotoxicity studies a second mammal species (rabbit) is required. This is due to a different metabolism of rodents: For instance, the organ toxicity of thalidomide could not be detected in developing offspring of the rodent species (rat). However, there are also limitations in rabbits: They often lack kinetic and toxic data and are vulnerable to some antibiotics and disorders of the digestive tract. In addition, clinical signs can be difficult to interpret. (51).
Rabbits are used for vaccine development. During the production of many monoclonal antibodies, non-human primates (NHPs) are the only pharmacologically relevant species (see ICH S6(R1) (52). NHPs are only used if no other relevant animal species is available. However, scientists are trying to replace NHPs with genetically modified mice (52).
Regulation mills grind slowly
In October 2017, the European Medical Agency hosted a workshop on "non-animal approaches" with the aim of revising the ICH guideline S5(R3). The European Medicines Agency (EMA) as regulatory authority appears to be cautiously open-minded towards "alternative assays" (53), which should become part of an integrated test strategy. However, "alternative assays" here do not only mean in vitro, but also ex vivo and non-mammalian in vivo assays, such as cell systems, tissue, organs or organisms (e.g. whole embryo culture with rat, rabbit and zebrafish embryos). Some of these assays are currently being investigated under defined scenarios for regulatory use. The assays are regarded as an important contribution to the evaluation of ambiguous animal test results. The revision of ICH S5(R3) under consideration of these new methods it is not expected that the animal experiment will end per se, but that under specific circumstances a complete replacement of one of the two animal species will be possible. However, no centralised approval process for the regulatory recognition of qualified alternative assays is planned to be initiated; this would remain "agency-specific" (53).
Literature:





 Dr. rer. nat.
Dr. rer. nat. Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.
Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.