Most of our organs are protected against harmful influences by barriers. This does not mean that no substance or signal enters the organ, but that nature has established certain mechanisms to protect the organism from external influences. This happens on the skin as well as on the cornea, on the internal organs such as the intestines or lungs and at the transition to the brain or between placenta and fetus. Researchers like Prof. Claus-Michael Lehr and his team as well as the conference participants are investigating such barriers in order to be able to overcome them if necessary with active pharmaceutical substances or, in contrast, to restrict their effect.

Biological Barriers Congress in the lecture hall of the Pharmaceutical Faculty of the University of Saarbrücken.
Photo: HIPS.
The conference participants are researching ways to make the "journey" of pharmaceutical molecules more effective, by using a spray, gel or in the form of a therapeutic antibody. However, in many cases, far too little active substance arrives in the tumor to be combated - a fatal development for the patient.
Therefore it is a matter of course for most of the researchers to work with cell cultures and - if it is necessary - to develop increasingly complex three-dimensional tissue cultures to simulate the barriers on a miniature scale. Such barrier research does not usually require an animal model. It is locally restricted. In order to understand the mechanisms all other factors must be excluded. The exact mechanism of cells and their components such as receptors, signal transduction pathways, gene regulators, microRNAs or extracellular vesicles in reaction to a foreign substance is required here.
Therefore, on the one hand, increasingly complex intestinal, lung or skin models, placenta and blood-brain barrier cultures as well as in vitro disease models - active pharmaceutical substances should ultimately benefit the sick people - played a major role in the conference. On the other hand, nanomedicine was a big topic, which should contribute to overcoming the barriers or targeted placement of the active substance. Another important topic was the establishment of bacteria in in vitro models.
At this point only a few examples of research results can be presented here.
Prof. Heike Walles and her team from the University of Würzburg have already developed a miniaturised blood vessel system for their skin and intestinal models as a standard. The team has also developed lung, colon and breast cancer models to develop monoclonal antibodies for the cancer therapy. Integrated bacterial cultures, e.g. in infection models, are used to investigate how bacteria succeed in penetrating the barrier. Various lung and nasal mucosa models are used to "load" nanoparticles with mucolytic drugs and to examine how the mucus is digested. Human skin models are also used to study the development of the TseTse fly. The parasite only affects humans and triggers what is known as sleeping sickness.
Dr. Kathrin Ribbeck from MIT in Boston focused on the function of mucosal barriers against infections. She found out that the mucosal barrier does not prevent the attachment of bacteria, but instead interferes with the biofilm formation of bacteria. She has also studied the cervical mucosa. There are cases where poor mucosal quality leads to the rise of bacteria into the uterus during pregnancy, which can cause premature birth. A microfluidic organ-on-a-chip system here is used to investigate the transport of bacteria through the mucosa.
Dr. Rana Abu-Dahab has developed an in vitro model of cystic fibrosis. Lung epithelial cells and sputum serving as mucosa were cultivated here together with the test bacterium Pseudomonas aeroginosa, a bacterium known as hospital germ. Then, an antibiotic was tested with the model. The researcher was able to show that the lung cells survive better if the mucus had previously suppressed the bacterial biofilm. Justus Horstmann of HIPS has developed a similar model.

Young scientist Justus Horstmann from HIPS presents his development of a cystic fibrosis model.
Photo: Christiane Hohensee
Another topic was tuberculosis, on which, according to the Lung Information Service, 10.4 million people worldwide were suffering in 2016. 1.7 million died. Maximilian Guerterrez from the Francis Crick Institute used an in vitro model to show that the tuberculosis pathogen surrounds itself with a lipid layer. Unfortunately, drugs interact with this lipid layer, but not with the lung pathogen itself. This leads to treatment failures. In the organism the pathogen hides in lipid droplets of macrophages.
Prof. Josué Sznitman of the Israel Institute of Technology in Haifa considers the current in vitro lung models to be too large (2). Conventional models with isolated lung cells and simulated air-liquid boundaries through submerged cell culture or transwell inserts with permeable membranes are also viewed critically. In his opinion, the models must create a realistic physiological respiratory environment through realistic anatomy of the respiratory tract, respiratory air flows and a cellular construction principle. Microfluidic lung-on-a-chip systems could be helpful here. Instead of a lobule, he and his team have developed a so-called acinus on the chip. An acinus is the smallest functional unit of an end portion of the lung. 12 azinus form a lobule.
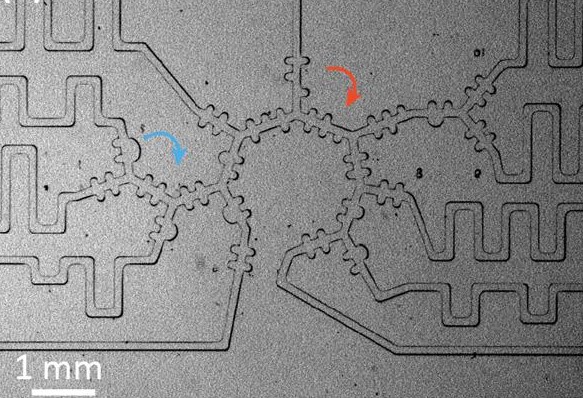
Respiratory tree of the alveolus in a microfluidic system. In a microfluidic model for the development of airways at the level of the acinar, saccular alveolar spaces are created (blue arrow) and subalveolar azine channels (red arrows). Accordingly, the air and liquid flows are dynamic, e.g. with turbulence.
Source: Tenenbaum-Katan et al. Biomicrofluidics 12, 042209 (2018). Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/). https://doi.org/10.1063/1.5023034
The young scientist Gianpiero Lazzari from the Institute Galien of the Université de Paris-Sud has developed a complex pancreas model in a microfluidic system. It consists of fibroblasts, pancreatic cancer cells and tiny blood vessels constructed in a matrix (3). The tumor model could be used for research into therapies based on nanomedicine.
Dr. Tina Buerki-Thurnherr from the Swiss Federal Laboratories for Materials Science and Technology (EMPA) is using placenta organoids on the chip to investigate whether nanoparticles can overcome the placental barrier. Nanoparticles as potential sources of environmental damage have not yet been sufficiently investigated. The researchers assume that exposure to nanoparticles during pregnancy could have a damaging effect on the fetus. These particles, ranging in size from 1 to 100 nanometres, are an integral part of our lives and constantly surround us. For example, they are contained in toothpaste as cleaning agents. The placental model is suitable for penetration studies.
Dr. Joanna Reyman from the Tübingen-based company CureVac is also investigating how barriers can be overcome. Using a human lung cell model, she is investigating how it might be possible to use a so-called messenger RNA as a kind of ferry to deliver a desired drug to the lungs. It is not so easy, because extracellular and intracellular barriers usually prevent foreign substances from getting in here. Messenger DNA occurs naturally in the cell during protein biosynthesis and is therefore of interest as a drug supplier in the cell.
Prof. Jesús Pérez-Gil from Complutense University in Madrid is investigating the surfactant on the surface of the lung. About 90% of the substance formed by pneumocytes of type II consists of lipids, the residue of proteins, which also include plasma proteins. A certain plasma protein is of interest to him because it "snitches" foreign substances to macrophages with its surface domains. It therefore has an immunological function because the nanoparticles, which are supposed to carry drugs through the lung barrier as carrier particles, are then fought or digested by the macrophages.
A exciting topic is the characterization and, if necessary, the use of extracellular vesicles as drug delivery systems. Extracellular vesicles are membrane particles that are formed by every cell and can be absorbed by a wide variety of cells. They can easily penetrate biological barriers and carry proteins, DNA or microRNA. Thus, they play an important role in intracellular communication in the tissue. The exact mechanism is not yet sufficiently known. Depending on their size, scientists distinguish between exosomes, microvesicles and apoptotic bodies. Dr. David Carter from Oxford Brookes University is investigating the role of exosomes in the stress response of cells. He found that irradiation also affects neighbouring cells, although they had not been exposed to stress at all. The background is that the neighbouring cells take up exosomes with RNA from stress cells.
However, extensive research is still required before extracellular vesicles can be used as drug delivery systems.
The examples clearly show that for the development of new required therapies, e.g. with the help of nanomedicine, the detailed mechanisms of action can only be researched with the help of human-specific, physiological cell and tissue systems.
For more information:
(1) http://biobarriers.hips-wordpress.helmholtz-hzi.de/
(2) Janna Tenenbaum-Katan, Arbel Artzy-Schnirman, Rami Fishler, Netanel Korin & Josué Sznitman (2018). Biomimetics of the pulmonary environment in vitro: A microfluidics perspective. Biomicrofluidics 12, 042209.
(3) Gianpiero Lazzari, Valérie Nicolas, Michiya Matsusaki, Mitsuru Akashi, Patrick Couvreur & Simona Mura (2018). Multicellular spheroid based on a triple co-culture: A novel 3D model to mimic pancreatic tumor complexity. Acta Biomaterialia.
(4) Pharmazeuten treffen sich zur internationalen Konferenz „BioBarriers 2018“ in Saarbrücken. Pressemitteilung der Universität des Saarlandes vom 02.08.2018. https://idw-online.de/de/news700215.
(5) Holger Büchner (2018). "Wir sehen Potenzial, von Tierversuchen wegzukommen" - Ein Gespräch des Saarländischen Rundfunks mit Prof. Claus Michael Lehr, Experte für Biopharmazie und Pharmazeutische Technologie am Helmholtz-Zentrum für Pharmazeutische Forschung im Saarland (HIPS), 27.08.2018. https://www.sr.de/sr/sr2/themen/politik/20180827_prof_claus_biopharmazie_tierversuche_interview100.html.
(6) Arbeitsgruppe im Portrait: Helmholtz-Institut für Pharmazeutische Forschung Saarland (HIPS), Abteilung Wirkstoff-Transport. https://www.invitrojobs.com/index.php/de/forschung-methoden/arbeitsgruppe-im-portrait/item/3222-arbeitsgruppe-im-portrait-helmholtz-institut-fuer-pharmazeutische-forschung-saarland-hips-abteilung-wirkstoff-transport




 Dr. rer. nat.
Dr. rer. nat. Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.
Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.