Across Barriers, a spin-off company from the Saarland University, Faculty of Pharmacy, was founded in 1998 by Prof. Dr. Claus-Michael Lehr and Dr. Eleonore Haltner. It employs 36 staff members. The company works closely with the Saarland University.
Headquarters of Across Barriers in Science Park, Saarbrücken.
Photo: Across Barriers.
Across Barriers aims to develop new in vitro methods for the characterization of active pharmaceutical ingredients providing them as a service for pharmaceutical, cosmetic and chemical industries. The company is certified according to GLP (Good Laboratory Practice)1, that means its services in the field of non-clinical laboratory work are certified as being of a high international standard. The company is also GMP certified (Good Manufactoring Practice)2, which is of great importance for the pharmaceutical, chemical and cosmetic manufacturers. Since 2009 the company has been approved for the American market by the Federal Drug Association.
The company was honoured by several prizes such as the "Founder Champion 2000" from the Federal Ministry of Economics and the "Entrepreneur of the Year 2002", awarded by Ernst & Young.
Serving the pharmaceutical or chemical industry, Across Barriers characterises pharmaceutical substances physico-chemically, that means it examines parameters of the test substance, such as fat and water solubility, acid dissociation constant in water, protein-binding capacity and stability of the substance. They also research the properties of molecules (1).
Moreover, the metabolism of the test substance is studied in vitro in cell cultures or artificial tissue. For this purpose, the company has its own research and development department and develops innovative in vitro test systems which simulate transport of substances and formulations through barriers and serve as a replacement to traditional animal tests.
Depending on the application site the substances need to traverse various barriers. Here, the skin, nose or mouth, the cornea of the eye, the mucosa, the bronchial or lung barrier or the blood-brain barrier can be involved. On the one hand it has to be examined whether and how the investigated compounds penetrate the barrier and, whether and how they are altered and / or transported. Since, for example, an oral intake of drugs can cause side effects, and the substances are removed by the liver, or at least changed before they arrive at the site, researchers develop test systems to avoid this problems. For example they might create an artificial cornea to test on. Such test systems are not just important for the medical industry but also of great importance for the cosmetic industry.
In order to investigate metabolism and other processes such as the penetration of substances in detail, human-specific cell or organ culture systems or artificial organs can be used. The company Across Barriers works on the development of such systems.


Also of great importance is the training and development of Across Barrier´s staff.
Photo: Manuel Sacha.
Across Barrier´s skin-bank and skin models
Since the skin of animals differs from the skin of humans in essential features and since such test results would only be of limited transferability to humans, Across Barriers has developed a skin bank, in which human skins from cosmetic surgery are stored. For substance tests, human skins of various thickness, e. g. full-thickness skin in vitro models, sections of a thickness of 300 to 500 micrometers, only one skin layer or the top layer of skin, the stratum corneum.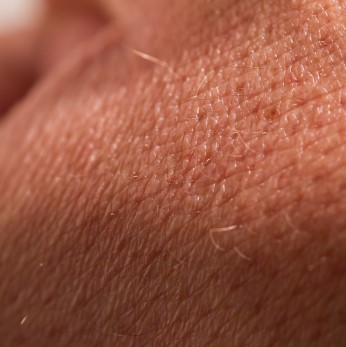
Human skin: The origin for human in vitro skin models and the Across Barrier´s Skin Bank.
Photo: Antorti, iStockphoto.
However, other human in vitro skin models are used such as EpiDerm (patent by MatTek) or EpiSkin (SkinEthic Laboratories, France). They enable examination of permeation (transport of substances to an acceptor region) or penetration (penetration into the various layers of skin, which serve themselves as acceptor regions). For permeation tests, the skin is clamped in a Franz diffusion cell3, for penetration testing the so-called "Saarbrücken" model4 is used. After an exposure time, the skins´ layers are cut with a microtome device (a cutting machine, with which one can disect micrometer-thin slices) followed by an analysis via UV detectors, by liquid chromatography or by mass spectrometry to find out where, and how much of the active substance passes into the individual layers. The permeation test is also concerned with the rate at which a substance penetrates into the skin layers.
Gastrointestinal cell culture model with Caco2 cells
After oral intake of a substance such as a drug, the intestine acts as a major barrier to entry into the bloodstream. In order to examine permeability, absorption and prediction of bioavailability and toxic effects of the substance, an in vitro cell culture with Caco2 cells is used. This cell culture was first isolated from a human colon adenocarcinoma and characterized by Dr. Jorgen Fogh from the Sloan-Kettering Institute for Cancer Research, New York, in 1977 (2). Since then it has often been optimized and standardized, with significant contributions of Dr. Eleonore Haltner and Prof. Claus-Michael Lehr (3, 4, 5, 6).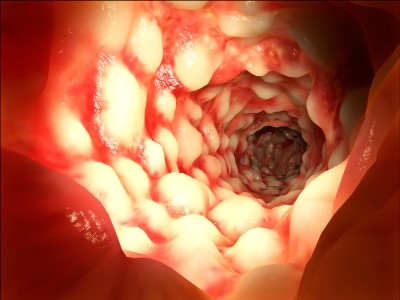
View of the small intestine with Crohn's disease, a chronic inflammatory bowel disease. For testing new drugs against this disease, cell culture systems with Caco-2 cells are used.
Photo: Selva Negra, iStockphoto.
Across Barriers uses the cell culture systems for permeability screening tests and for studies to classify a drug according to the biopharmaceutical classification system (BCS)5. The BCS classification is required by both by the U.S. FDA regulations as well as by the European Medicines Agency test guidelines, in order to evaluate the bio-equivalence6 of drugs.
Alternatively, pig intestines from slaughtered animals are tested using the Ussing-technique7.
Artificial lung model
The artificial lung model consists of Calu-3 cell cultures grown as monolayers (single cell layers) on a permeable membrane in small removable plastic trays called transwell inserts. They are supplied with nutrients dissolved in water.
There are two ways to supply nutrients:
1) the entire cell layer is under water (the traditional so-called LCC culture), from which the nutrients reach the cells from the top and the bottom
2) the modern air-liquid interface culture, wherein the top of the culture is exposed to air.
The latter simulates the flow in the human lung better because they develop a layer-like growth habit, the cells show more cilia and mucus which is more typical of these cells. The cells take 10 days before they have formed cell-cell junctions - called tight junctions - and are ready for application. The tight junctions represent the real barrier for substances in lung tissue. As would also be the case in the lungs, the inhaling substance meets the top of the cells via the air and then has to pass through the barriers. With its cell culture, Across Barriers investigates the permeability kinetic (how fast a substance can permeate into the lung cells and what the concentration dependency is), the efflux (outflow of the substance from the cell) or, whether a specific protein (P-glycoprotein8) is inhibited by the test substance. The protein transports toxic substances from the body, however it also binds to drugs, which can then no longer be effective.
6-Well plate with five inserts.
Photo: Christiane Hohensee
Artificial cornea model
The latest development is an artificial cornea model consisting of human cells that was presented at the Congress on Alternatives to Animal Testing in Linz, Austria.
Until now, large numbers of animals have used to test new drugs for treating eye diseases, and in order to investigate the influence of excipients in ocular medication on the cornea. The in vitro model, also called “human cornea construct” (HCC), was made using immortalised human corneal epithelial cells (HCE-T cell line) and a human cornea keratinocyte cell line (HCK Ca cell line).
The model has already been optimised, evaluated and pre-validated in several laboratories (7, 8). It examines the permeability and the tolerability of the drug, including the compatibility of preservatives. The model has a three-layered structure with a multilayer epithelium that emulates the main barrier properties with regard to the permeation of substances through the cells.
----------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
The HCC’s structure is the result of a gradual build-up in special culture vessels, which enable both submerged cultivation in a culture medium as well as cultivation at the air-medium boundary. Here, a polycarbonate filter is coated with a cellular collagen gel on which the endothelial cells are seeded. The endothelial monolayer is overlaid with collagen gel-containing stromal fibroblasts and then submerse cultured until confluence (densest possible structure) is achieved. The level of the medium is then lowered somewhat and the constructs raised with the aid of a metal plate. After several days of culture at the air-medium boundary, a multilayered epithelium develops. The complete cultivation of the HCC takes four weeks (9, 10).
-----------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
The HCC model is made using transformed human cell lines are used. For this purpose, the cells are usually “transfected” with an SV40 virus, or the corresponding gene for the SV40 large T antigen is inserted. As a result, the cells are almost indefinitely cultivable (immortalised), without losing their characteristics (9, 10).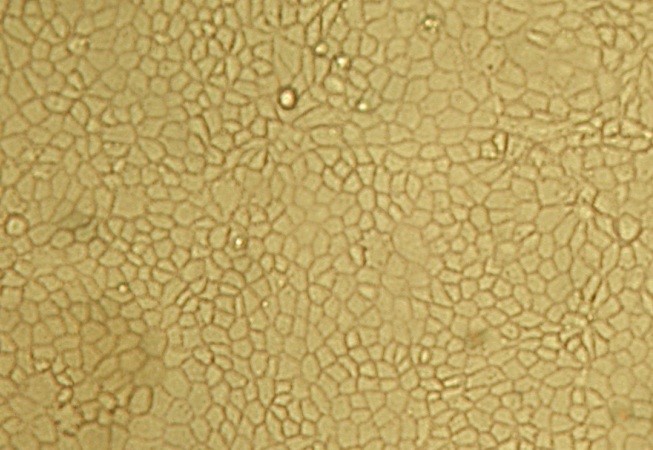
HCE-T corneal epithelial cells, 95 per cent confluent (20x magnification).
Photo: Gustavo Guzman, Across Barriers.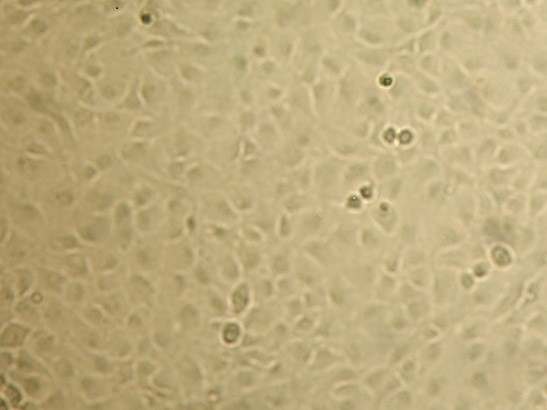
Cornea keratinocyte cell line: HCK-Ca, 95 per cent confluent cells (20x magnification).
Photo: Gustave Guzman, Across Barriers.
In order to collect data on the permeability of the developed cell culture, the so-called transepithelial electrical resistance (TEER)9 is measured. It is a good biomarker, as for example in an inflammatory process the resistance is reduced and the barrier becomes more permeable. In this case compatibility and incompatibility of the active ingredient and the admixed preservative were stated (7).
Blood-brain barrier
When applying medication to the central nervous system, agents have to pass the blood-brain barrier to be active at the target site. In contrast to water-soluble substances, fat-soluble substances are able to pass through the blood-brain barrier and subsequently from the blood to the brain. With the help of carriers (channel proteins) in the membrane, the blood-brain barrier is also actively involved in the metabolic process, because transported molecules are chemically modified (11). Across Barriers has established and standardised an in vitro model for investigating the permeability of substances across the blood-brain barrier. It is based on porcine cerebral capillary endothelial cells, which is why it does not yet represent a complete replacement procedure for animal experiments. It is intended to act as a link between data from animal experiments and computer prediction models. Since the blood-brain barrier is not easily passed, transport studies are of great importance. Not only substances which are meant to pass the blood-brain barrier can be tested, but also those which must be prevented from passing it. The cells are cultivated in a single layer on a polycarbonate membrane in a transwell system. After seven days, the test substance can added. The permeability is analysed and quantified using the high performance liquid chromatography technique (HPLC)10.
Artificial fingernail model
The artificial fingernail model is used to identify potential drugs, such as those developed for treating nail fungus. Fingernails are made up of a special kind of keratin and not living cells, so that they currently cannot be grown artificially with the necessary characteristics. Since human fingernails are not available in sufficient quantities, the scientists have developed a model that uses skin of bovine hoofs from slaughterhouses.
All in vitro models have the goal of replacing animal experiments and, where this is already technically possible, using human-specific material.
InVitroJobs interviewed the managing director and founder of Across Barriers, Dr. Eleonore Haltner-Ukomadu.
InVitroJobs: Across Barriers – how did you come by the name?
Dr. Eleonore Haltner-Ukomadu: The name is derived from our work. The work done by Across Barriers is research into the transport of pharmaceutical ingredients through biological barriers. When we had the idea for the name, we did a small survey of students at the university. Students from the area of natural sciences and pharmaceutics draw the right conclusions from the name, associating it with absorption. Students from areas other than natural sciences associated the name with human rights organisations and for instance with communication, which was important for us. In all cases the name was perceived as positive and did not convey any negative associations.
InVitroJobs: What was the founding period like in a company specialising on replacement methods for animal testing?
Dr. Eleonore Haltner-Ukomadu: It was difficult at the start, because the methods we were working with hadn’t officially been approved. That means that the methods were used cautiously by industry, but only published in research reports. Therefore, something had to be done to achieve acceptance of the methods.
InVitroJobs: You work within three GLP categories. Which categories are they?
Dr. Eleonore Haltner-Ukomadu: The three GLP categories are 1, 8 and 9. This means approval for analytic work, physico-chemical tests and in vitro tests based on cell and tissue tests.
InVitroJobs: The company was strongly involved in the standardisation of the Caco-2 cell lines. What were the most important stages and what can you say about the development?
Dr. Eleonore Haltner-Ukomadu: The most important stage was first of all to try and develop a standardised test design in order to be able to obtain reproducible data in our own lab with little standard deviation. The second step was to conduct the cell qualification as recommended by the FDA (BCS, Biopharmaceutics Classification System5) using 19 substances in accordance with FDA regulations. After that came the cooperation with other companies and also the FDA, in order to ascertain how well the cell model can be transferred from one lab to another and how reproducible the data are between the various labs. Different influencing factors were determined, such as the influence of the buffer or the pH value. We also determined that it is not the absolute values of the measured data that can be compared, but that the comparison must be made by means of a so-called calibration curve, such as is now also defined in the FDA’s BCS Guideline.
InVitroJobs: Die Caco-2 cells originate from the large intestine, so what makes them suitable for studying penetration or absorption of the small intestine? After all, the cells in the small and large intestines have different tasks and must therefore also have different receptors.
Dr. Eleonore Haltner-Ukomadu: That is pure coincidence. The Caco-2 cells were discovered in 1947 by Fogh. Mr Fogh studied more than 100 different intestinal cancer cells and thus also discovered the Caco-2 cells. A later characterisation showed that, although they originated from a colon carcinoma, they are much more similar to enterocytes in the small intestine. This means that they form a dense mono-layer and express transport systems also expressed in the small intestine. For this reason, Caco-2 cells have also become an established cell model for studying absorption from the small intestine.
InVitroJobs: Isn’t it difficult converting bowel cancer cells such as the Caco-2 cells to reliably working cell cultures? After all, they used to be degenerated cells.
Dr. Eleonore Haltner-Ukomadu: The advantage of using a cancer cell line is that you don’t have to convert it any more. It already has certain properties and need not be further transfected (Transfection: introduction of foreign DNA or RNA into eukaryotic cells. Ed.) in order to immortalise them, as they are already immortal and can easily be cultured in the lab. Of course, the Caco-2 cell line behaves like any other cancer cell line, changing relative to the duration of cultivation. Determining the influencing factors that lead to changes, and conducting the culture correspondingly, is therefore part of the standardisation of a cell model based on cancer cells. An example can illustrate this: Under some culture conditions, Caco-2 cells lose their original characteristics after 40 weeks. Therefore we grow a new culture every 35 weeks, completely characterise it and only then use it in tests again.
InVitroJobs: What is the current stage of development? Can you estimate whether and when the next model may be validated?
Dr. Eleonore Haltner-Ukomadu: The ocular model was prevalidated within the framework of a BMBF (German Federal Ministry of Education and Research. Ed.) project according to the relevant ECVAM standards. We already use the model for studies in the field of research and would very much like to validate it in accordance with the corresponding regulations. However, validation studies are extremely expensive and are not normally funded. That means you don’t get any financial support from either the BMBF or EU project funding to conduct such costly research. It is therefore very unlikely that we be able to validate the model with partners in accordance with the regulations, because the costs cannot be carried by either small to medium-sized enterprises or universities. We are proceeding with this model exactly the same way as with the Caco-2 model, which has never officially been validated, in that we continue to work with the model, conduct further research on reproducibility and conclusiveness at our own expense, then publish the results and hope that this way we can gain the acceptance A) of our customers and B) of the authorities.
InVitroJobs: At present collagen from rats is still used for producing the collagen gel into which the human cornea keratinocytes are introduced. Will there be no substitute from human material in the foreseeable future?
Dr. Eleonore Haltner-Ukomadu: On the one hand it would certainly be desirable to have an entirely human model and thus also to replace the animal collagen with human collagen. On the other hand this approach might have the disadvantage of such human material being less readily available. If a model is to be implemented worldwide as a replacement for commonly conducted animal tests, it is also important to have all the model’s components, such as the collagen, in sufficient quantities and consistent quality. This is always difficult in the case of human material and also pushes ethical boundaries. It would be far more interesting to try and replace collagen structures with other gels, either plant-based or on a polymer basis, because these materials would be available in large quantities.
InVitroJobs: How can the problem be solved that the Draize test also involves testing the conjunctiva for inflammation or damage?
Dr. Eleonore Haltner-Ukomadu: That is still completely unresolved, that would be the next step. The model is constructed for investigating the permeation of pharmaceutical active agents and irritation of the cornea. The first task would be to clarify whether the conjunctiva comprises similar cell types and therefore this model can be employed, or whether a further model for the conjunctiva must be constructed.
InVitroJobs: Is the development of a blood-brain barrier model using human endothelial cells conceivable? Are you working on that?
Dr. Eleonore Haltner-Ukomadu: Culturing endothelial cells is very difficult. At the moment we obtain the endothelial cells from the capillaries of porcine brains, as pigs are killed for other reasons anyway. It is absolutely necessary to obtain the capillaries from the brain while they are still fresh, i.e. there can only be two hours between killing the animal and obtaining the endothelial cells. This means it is entirely impossible to build such a model based on primary cells using human material. The only possibility might be to use stem cells and differentiate these so that they behave like cerebral capillary endothelial cells.
InVitroJobs: In some of the in vitro methods you still use animal cell or tissue material, such as primary alveolar cells from pigs. Is this demanded by your clients? Do you plan to gradually shift to completely using human-specific tissue?
Dr. Eleonore Haltner-Ukomadu: On the one hand that would be very desirable, as we are conducting experiments with substances intended for use on humans. On the other hand shifting to human models always poses the problem of tissue availability. This makes it very difficult to replace tests when there is always a recurring shortage of tissue. Very often the required cell type cannot be isolated from the tissue from corpses. One possible option for the future could be to establish such models using stem cells. At present, human cell models often involve the use of biopsy material, but here too we are faced with the question as to how representative this material is for a healthy human.
InVitroJobs: What experience do you have with respect to comparisons between cell and tissue cultures of human or animal origin?
Dr. Eleonore Haltner-Ukomadu: We already have compared models based on animal cells with models based on human cells. One example is the alveolar cell – i.e. lung – model. We at Across Barriers work with porcine alveolar cells, and the chair of my doctorate supervisor Prof. Lehr has worked in the past with human lung cells obtained by biopsy. We have compared the absorption of both models and could not determine any difference.
InVitroJobs: Some of your in vitro models pertain to the endpoint “repeated dose”, are thus also relevant to the decision regarding maintaining a ban on cosmetics tested on animals. This study goal ought to be the hardest to find sufficient replacement methods for. What do you think about that?
Dr. Eleonore Haltner-Ukomadu: It is indeed true that it is very difficult conducting repeated dose investigations using in vitro models based on tissues or cells. Many cell culture models only have a lifetime of a few days and therefore cannot be used for repeated dose studies. That goal still involves a great deal of time and effort. There are, however, already examples of long-term cell cultures in which cells could be cultured for between several months and a year without losing their characteristics. One such example is lung cells. Our company is also working on projects aimed at achieving this. One approach is to establish cell cultures in microfluidic chips, because these systems allow the generation of the necessary shear forces and also because the nutrition status of the cells is much better. The assumption is that this facilitates prolonging the life duration of the cells, which is the basic prerequisite for being able to conduct repeated dose studies.
InVitroJobs: Thank you for the interview.
Glossary:
1 Good laboratory practice (GLP): The OECD principles are a quality control programme for harmonising test methods. To create a foundation of trust for the mutual recognition of test data, it must be ensured that the laboratories conducting the tests have the same standards in all countries and work according to similar methods. A lab or a method can be GLP certified within this context.
2 Good Manufactory Practice (GMP): A good manufacturing practice (GMP) is a production and testing practice that helps to ensure a quality product. Many countries have legislated that pharmaceutical and medical device companies must follow GMP procedures, and have created their own GMP guidelines that correspond with their legislation. (http://en.wikipedia.org/wiki/Good_manufacturing_practice)
3 Franz cell or Franz'sche diffusion chamber: Franz diffusion cells are a two-compartiment in vitro test system designed to study drug diffusion. It constists of a donor and a acceptor compartment, separated by an artificial membrane artificial, reconstructed skin or excised skin. Drug formulation is applied in the donor compartment, samples are taken from the acceptor compartment i npre-determined time intervals.
4 Saarbrücken model (Saarbrücken Penetration model): with this apparatus, the drug amount within the stratum corneum can be measured in single corneocyte layers using tape strips which extract the drugs from the skin layer. Than the drug can be scanned on the tape. Unnaturally hydration of the skin can be avoided, the skin itsself is the acceptor compartment.
5 Biopharmaceutical Classification System (BCS): drugs are classified into four groups depending on their solubility and permeability (http://www.fda.gov/AboutFDA/CentersOffices/OfficeofMedicalProductsandTobacco/CDER/ucm128219.htm)
6 Bio equivalence: The term derives from the pharmacokinetics and assess the interchangeability of two drug-drug equivalent, differing only in the manufacturing process and / or the information contained adjuvants (http://de.wikipedia.org/wiki/Bio% C3% A4quivalenz).
7 Ussing technique: an ussing chamber is an electro physiological apparatus for a long-term cell culture of epithelia and measurement of barrier ans transport functions of living tissue (http://www.fh-jena.de/~gitter/ussikamr.pdf)
8 The P-glycoprotein has an important function for body protection because it transports potentially toxic substances out from the active cell. In this way it can also prevent the effective treatment of diseases, as many drugs are ligands of P-glycoprotein. (Http://www.pharmazeutische-zeitung.de/index.php?id=41568)
9 transepithelial electrical resistance (TEER): The barrier function is determined by the measurement of the electric resistance above the epithelia (transepithelial resistance = TER). The resistance in relation to the epithelia area. It is defined in unit Ohm x square centimeter). I fthe epithelia transports ions actively from one site to the other (electrogenic transport), a transepithelial current is generated. (http://www.fh-jena.de/~gitter/ussikamr.pdf)
10 HPLC (High-Performance Liquid Chromatography): is a chriomatographic technique used to separate a mixture of compounds in analytical chgemistry and biochemistry with the purpose of identifying, quantifying and purifying the individual components of the mixture. (http://en.wikipedia.org/wiki/High-performance_liquid_chromatography)
Literatur/Quellen:
(1) http://www.uni-saarland.de/fak2/komm/_doc/profil_acrossbarriers_en.pdf
(2) Braun, J. S. (2007): Arzneistoffabsorption in Caco-2/TC7 Zellen: Ibuprofen und derMonocarboxylat-Transporter 1 (MCT1). Dissertation an der Universität Tübingen.http://tobias-lib.uni-tuebingen.de/volltexte/2007/3033/pdf/Dissertation_Jutta_Braun.pdf
(3) Haltner, E., Schmitz, S., Gindorf, C. & Ruoff, J. (2001): In vitro Permeabilitätsuntersuchungen als Ersatz für Tier- und Humanstudien - welche Voraussetzungen müssen erfüllt sein? ALTEX 1/01 (http://www.altex.ch/en/index.html?id=50&iid=59&aid=31&PHPSESSID=54b804e9282565bb678e9ba279fd4952)
(4) Bock, U., Flötotto, T. & Haltner, E. (2004): Validation of cell culture models for the intestine and the blood brain barrier and comparison of drug permeation. ALTEX 2004; 21 Suppl 3: 57-64.
(5) Gindorf, C., Steimer, A., Lehr, C.-M., Bock, U., Schmitz, S. & Haltner, E. (2001): Markertransport über biologische Barrieren in vitro: Vergleich von Zellkulturmodellen für die Dünndarmschleimhaut, die Blut-Hirn-Schranke und das Alveolarepithel der Lunge. ALTEX 18: 155-164.
(6) Bock, U., Kottke, T., Gindorf, C. & Haltner, E. (2003): Validation of the Caco-2 cell monolayer system for determining the permeability of drug substances according to the Biopharmaceutics Classification System (BCS).(http://www.acrossbarriers.de/uploads/media/FCT02-I-0305_BCS.pdf
(7) Bock, U., Haltner, E., Guzman, G. & Reichl, S. (2012): Characterisation of Human Cornea Constructs for the Rapid Screening of Preservatives and Evaluation of Their Use in Permeability Assays to Reduce Animal Testing. Abstract zum Kongress für Alternativen zu Tierversuchen, Linz.
(8) Guzman, G., Reichl, S., Bock, U. & Haltner, E. (2012): Optimisation of Human Cornea Constructs For a Rapid Characterisation of Pharmaceutical Preparations, Excipients and Chemical Compounds. Abstract zum Kongress für Alternativen zu Tierversuchen, Linz.
(9) Reichl, S. & Müller-Goymann, C. C. (2004): Künstliche Hornhaut als In-vitro-Modell. http://www.pharmazeutische-zeitung.de/index.php?id=titel_20_2004
(10) Reichl, S., Bednarz, J., C C Müller-Goymann, C. C. (2004): Human corneal equivalent as cell culture model for in vitro drug permeation studies. Br J Ophthalmol. 88: 560–565.
(11) Bock, U. & Haltner, E. (2003): Porcine Cerebral Capillary Endothelial Cells to study Blood-Brain Barrier Permeability. http://www.acrossbarriers.de/uploads/media/FCT01-I-0305_BBB.pdf




 Dr. rer. nat.
Dr. rer. nat. Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.
Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.